The next big thing in cancer treatment? It’s small. Approximately 1/20,000th the size of a human hair small. And it’s being championed by Thomas P. Loughran Jr., MD, UVA Cancer Center director and Mark Kester, PhD, the new co-director of the UVA Institute for Nanoscale and Quantum Scientific and Technological Advanced Research (nanoSTAR). These two top minds are working together to reveal the potential of nanotechnology to treat cancer.
Supporting their efforts are the National Institutes of Health and National Cancer Institute, which bestowed a $10 million program project grant (PO1) for four projects utilizing nanotechnology in the treatment of acute myeloid leukemia (AML). “The idea is to use nanotechnology initially as a way of delivering cancer therapeutics and making it easier to get the drugs into patients, specifically inside the cancer cell,” says Loughran.
“There are plenty of great drugs that can eradicate cancer cells, but they also kill other cells, which results in all the side effects of cancer treatments,” says Kester. “If we put these drugs in little packages, then they go quickly through the body and can be delivered directly to the cancer cell.”
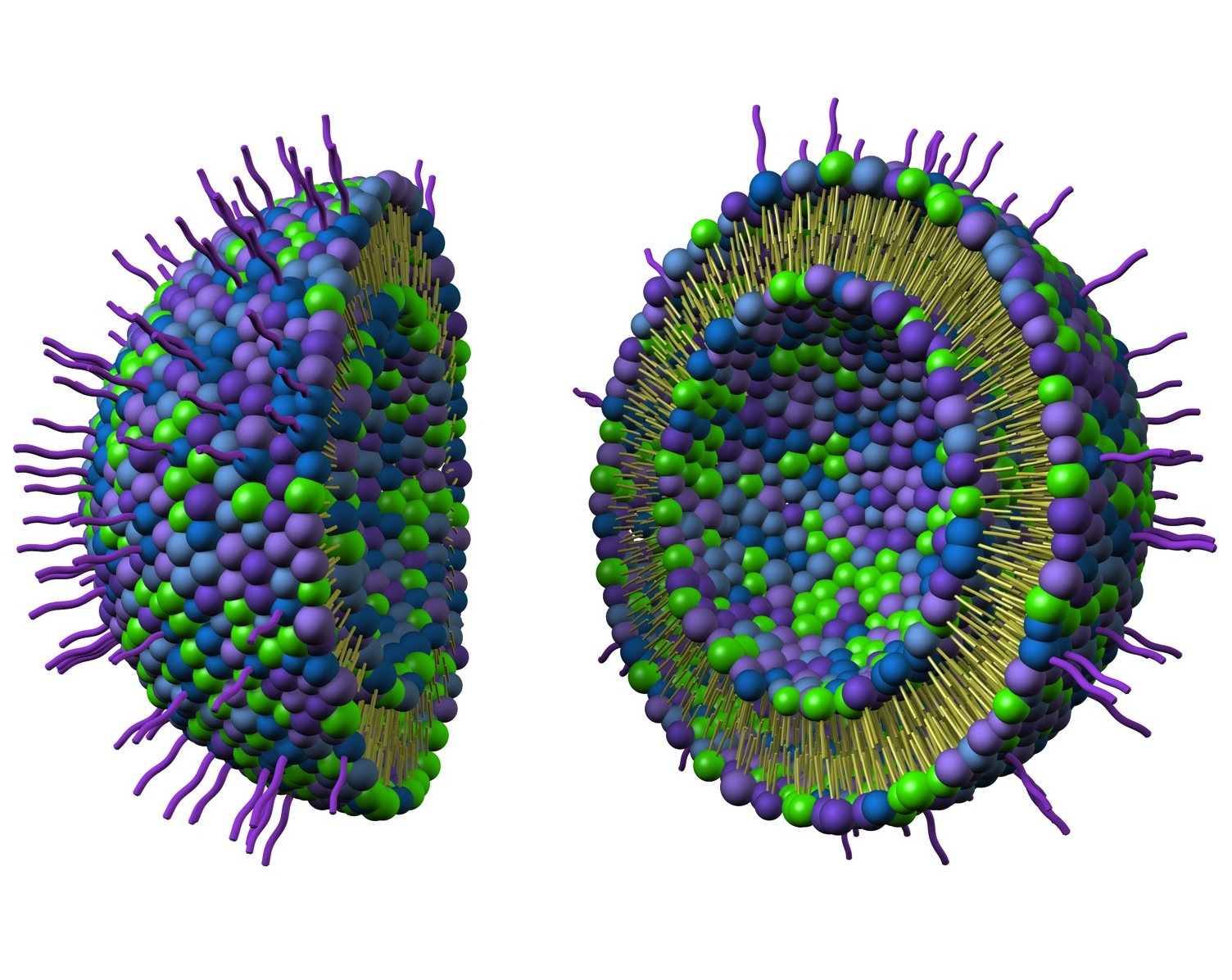
These “little packages” are the basis of nanotechnology. According to Kester, they are common materials manufactured as microscopic particles, which take on new properties that can then be exploited and used as a delivery platform. Among those currently being developed are nanoliposomes, or very small balls of fat.
A Breakdown of Nanoparticles
Well-accustomed to using analogies to help others visualize the way nanotechnology works, Kester describes the liposomes as small basketballs. “Instead of rubber, these basketballs are made of fat lipids,” he explains. “We can add in a lipid drug like ceramide that is selective, that can kill cancer cells but not normal cells.”
Inside the “basketball,” Kester says there is a void where they can add other drugs or chemotherapeutics that work in tandem with ceramide to more effectively eradicate cancer cells. Even those drugs that are too toxic on their own become viable when packaged in a nanoliposome. “Putting a highly toxic chemotherapy agent into a nanoliposome makes it nontoxic,” he says.
Basketballs have nubs that provide a better grip, and in this case, that layer is a protective shield of polyethylene glycol. “This keeps the particle from being eaten up in the body,” says Kester. “It stays in the body longer so it works longer.” Finally, the outermost layer is an antibody-targeting agent that tells the drug where to go in the body. “The pieces of the antibody bind to markers on the cancer cell like a lock and key,” says Kester.
Also possible is the addition of nanoparticles that allow for better imaging of disease. “Some nanoparticles enhance imaging of the tumor, so we can decipher it from regular tissue. We can then simultaneously seek out the tumor, image it and treat it,” says Kester.
Nanoliposomes are just one example. Other particles are being developed for the delivery of molecular-based drugs that will target specific gene mutations that cause an individual’s cancer cells to grow. It is the next phase in personalized medicine, according to Kester. “In three to four years, nanoplatforms could be used to deliver drugs targeting a specific gene mutation,” he says.
The Future Is Now
Seeing the potential of nanotechnology and actually putting it into practice may seem like a leap far into the future. However, this science is now being applied. Advancing it even further is a cause UVA is well equipped to handle.
“We anticipate that we will see in the next six months the first in-man clinical trials of this new technology, delivering ceramide for patients with liver cancer,” says Loughran. “This is outside the scope of our PO1, but it is a similar concept. If the trial goes well, we would do similar trial with AML. This is way ahead of the grant prediction. The purpose of the grant was not to develop clinical trials, but to develop the concept that this might be helpful for patients. We are now at the point where it is worth testing in patients because we are having remarkable success with leukemia in animal models.”
Foundation for Progress
As a renowned researcher and director of the Cancer Center, Loughran has worked to build a framework to support this type of advanced science at UVA, which includes investments in technology and new faculty and promoting collaboration among the schools. “In the bigger scope of things, this is an issue that spans the grounds,” he says. “This is a collaboration between the UVA Schools of Medicine and Engineering to benefit patients throughout the state.”
It is this foundation of excellence and collaboration that persuaded Kester to join the team. Kester was recruited from Penn State University, where he was director of the Penn State Center for Nanomedicine and Materials and the co-leader for Experimental Therapeutics at the Hershey Cancer Institute.
“What attracted me to UVA was the diversity of faculty across the UVA system that will allow us not only to develop but validate and commercialize the next generation of products used for cancer treatment,” says Kester. “The commitment to out-of-the-box approaches like nanotechnology is what defines UVA as a leader for cancer patients in Virginia.”