Half of men over age 60 have benign prostate hypertrophy (BPH) or enlarged prostate. For men who live to be 80, that number jumps to some 90 percent. As you’re likely well aware, this enlargement of the prostate gland causes lower urinary tract symptoms, such as frequent urination, painful urination, urgency, a weak urine stream and incomplete emptying of the bladder.
Half of men over age 60 have benign prostate hypertrophy (BPH) or enlarged prostate. For men who live to be 80, that number jumps to some 90 percent. As you’re likely well aware, this enlargement of the prostate gland causes lower urinary tract symptoms, such as frequent urination, painful urination, urgency, a weak urine stream and incomplete emptying of the bladder.
Because these symptoms can be embarrassing as well as inconvenient, a number of medical therapies have been developed to treat BPH, but all have drawbacks. Medications can cause side effects some patients may be unable or unwilling to tolerate, such as impotence and abnormal ejaculation. Surgical approaches — transurethral resection of the prostate and, in cases of the most enlarged prostates, open surgery — carry the potential for complications.
To provide men a potential alternative, UVA interventional radiologists Ziv Haskal, MD, and Andre Uflacker, MD, along with colleagues in the Department of Urology, are conducting an FDA-approved clinical trial of prostate artery embolization (PAE), a new interventional therapy that has shown potential for relieving BPH symptoms without side effects. They are currently enrolling up to 80 patients.
Setting the Standard of Care
Several medical centers in the United States, including UVA, now offer PAE after observing its successful adoption by European and South American hospitals. They can do so because PAE uses devices that the FDA has already approved for treating uterine fibroids in women. Indeed, the underlying principle—reducing the blood supply to the targeted tissue or organ, shrinking it and reducing symptoms—is identical. Interventional radiologists have also used similar techniques for years to treat prostate or bladder bleeding. However, because the procedure is still in its early stages, it still warrants this careful and detailed study, according to Haskal.
Haskal and Uflacker have chosen to conduct a one-arm rather than a randomized trial of PAE, which means that all patients who enroll will receive the exact same treatment they would have gotten as part of routine clinical care. “We are treating patients who meet uniform study criteria with a defined protocol that codifies the best procedures we would follow if we were treating these patients without a study,” Haskal says. “The advantage to the patient of being in an FDA-monitored, registered study is that patients receive very close clinical care and access.”
Although there have been a number of retrospective studies highlighting the efficacy of the new treatment, Haskal and Uflacker believe the best way to set the stage for PAE’s wider adoption is to conduct a prospective trial. “Retrospective studies tend to underestimate complications,” says Haskal, who as editor-in-chief of the leading peer-reviewed journal in the field, Journal of Vascular and Interventional Radiology, has reviewed many of these papers. “The upside benefit for men based on experience in Europe and South America seems quite positive,” he says. “The best way to prove this is through a prospective study.”
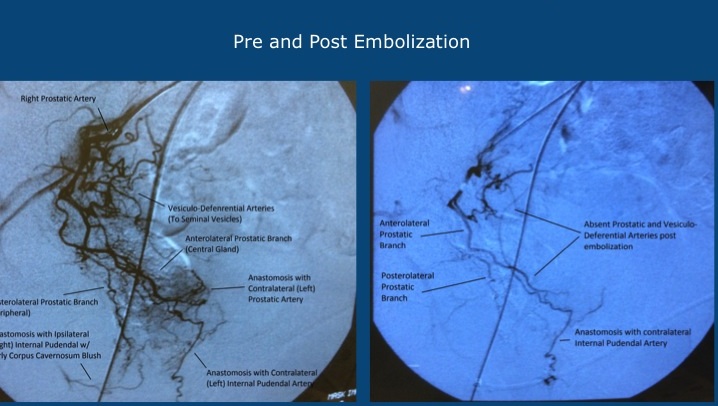
In PAE, doctors make a tiny nick in the groin and use a cone beam CT to guide a microcatheter to the end of the arteries supplying the prostrate. They then inject the appropriate number of microparticles needed to produce the desired reduction in blood flow. Patients are kept overnight and typically see their symptoms improve within a month. Learn more about the PAE procedure in the video below.
Although this approach is analogous in many respects to the first-line procedure for treating uterine fibroids in women, it does pose some unique challenges. “There is a great deal of individual variability in the anatomy and branching patterns in this area,” Uflacker says. “To ensure we select arteries that supply blood exclusively to the prostate, we are using sophisticated new embolization guidance software from Siemens during the procedure.” The use of this software highlights another goal of the study. “In addition to assessing the effectiveness of PAE based on alleviation of symptoms, we want to define the protocols that everyone can follow to maximize outcomes while minimizing risks,” Haskal says.
Study Eligibility
To be eligible for the PAE trial, patients should meet the following criteria:
- Age 45 to 80
- Six months of lower urinary tract symptoms diagnosed to be caused by BPH with or without acute urinary retention
- Refractory to medical therapy, defined as at least a six-month course of alpha-1-receptor antagonist and/or 5-alpha-reductase inhibitor. Patients in whom medication is not tolerated, refused or contraindicated may waive this requirement.
- Patients with acute urinary retention may have had a catheter for decompression of the bladder for any period of time prior to the initial evaluation.
Patients with the following conditions will not be eligible:
- More than two urinary tract infections a year, prostatitis or interstitial cystitis
- Biopsy-proven prostate cancer or urethral cancer
- Active penile cancer
- Lower urinary tract symptoms from other causes besides BPH
- Bladder sphincter abnormalities
- Surgical management in the past for benign prostate hypertrophy
- Long-term anticoagulation therapy that cannot be stopped for a procedure
To find out if your patient is eligible to participate in the PAE trial (IRB# 18236), contact study coordinator Brigitte Kelly, RN, at 434.297.7136 or bjk3c@virginia.edu.