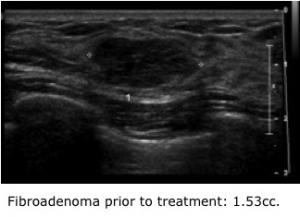
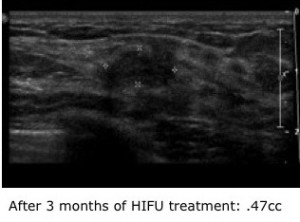
For women, the diagnosis that a lump within their breast is a fibroadenoma is good news. These common, benign tumors typically appear in young women between the ages of 15 and 35. Although they can usually be managed with simple observation, many women consider them bothersome and wish to have them removed. Their only option currently is surgery, which leaves a scar.
“We’re trying to give women a noninvasive alternative,” says breast surgeon David R. Brenin, MD, a co-lead investigator of the nation’s first clinical trial evaluating the safety and efficacy of high-intensity focused ultrasound to treat these tumors. “High-intensity focused ultrasound has been approved in Europe for fibroadenoma treatment. We would like to make this technology available to American women.”
An International Center for Research on Focused Ultrasound
Over the last five years, University of Virginia has moved to the forefront of institutions exploring the potential of focused ultrasound. With the help of $4 million in funding from the state of Virginia, UVA established the $8 million Focused Ultrasound Center in 2009, and clinical trials evaluating the use of focused ultrasound to treat disorders like Parkinson’s disease, essential tremor, uterine fibroids and other conditions are ongoing.
With focused ultrasound, an acoustic lens is used to concentrate multiple intersecting beams of ultrasound energy on a single point within the body with extreme precision and accuracy. The process is analogous to burning a hole in a piece of paper with a magnifying glass. “In essence, we are destroying the tumor from within,” says radiologist Carrie Rochman, MD, who has partnered with Brenin as a trial co-leader. “The goal is to reduce tumor size and consistency so that patients cannot feel it.” Thus far, Rochman and Brenin have recorded a 69 percent decrease in tumor size three months after treatment. Maximum benefit is expected to occur by the ninth month.
A Noninvasive, Outpatient Procedure
Rochman and Brenin are using an Echopulse® device, which enables them to map the fibroadenoma, review and modify the treatment plan suggested by the machine’s software and ablate the tumor, all in a single procedure. The Echopulse uses ultrasound to guide the procedure, rather than magnetic resonance imaging used in the UVA essential tremor trial.
Depending on the size of the fibroadenoma, the procedure can take between 30 minutes and two hours. Throughout, the patient is under conscious sedation with only local anesthesia. The Echopulse generates a series of approximately two-second sonifications with 30- to 40-second intervals in between to allow tissue to cool. “Patients typically feel the sensation of warmth during the procedure and minimal pain, if anything at all,” says Rochman. Patients can immediately resume normal activities once they have recovered from the sedation.
Patients in the trial will have a follow-up appointment with the breast surgeon after seven days, and a follow-up ultrasound and clinical appointment again at months three, six and 12 following the procedure. There are no costs for participating in the study.
The First Step Toward an Alternative Treatment for Breast Cancer
Once the trial is completed and if results warrant it, Rochman and Brenin hope to launch a multicenter pivotal study. If that trial is successful, Rochman and Brenin anticipate that ultrasound treatment for fibroadenomas will be approved by the FDA.
From there, Brenin believes this trial will set the stage for investigating the use of high-intensity focused ultrasound to treat malignant breast tumors. “Hopefully, if we can demonstrate success with benign tumors, we will be able to test the efficacy of this approach with malignant ones,” Brenin says.
Study Eligibility
The study is enrolling 20 patients who meet the following criteria:
- Participants in the study must be at least 18 years of age.
- Before being admitted to the study, patients will have a mammogram, diagnostic ultrasound and biopsy of the breast lump to determine that it is a benign fibroadenoma.
- The fibroadenoma has to be at least 1 cm in one dimension and must meet criteria regarding distance from skin and ribs that most fibroadenoma satisfy.
- Women who are pregnant, nursing or have a history of breast cancer in the same breast are not eligible.
To find out if your patient is eligible to participate in this study of high-intensity focused ultrasound as a treatment for breast fibroadenoma (IRB-HSR# 17277), please contact the study coordinator, Katie Rea, RN, at 434.243.0315 or uvastac@virginia.edu.
Read more about breast cancer screening and treatment at UVA.