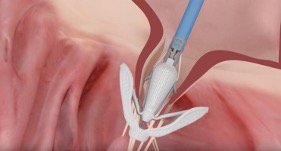
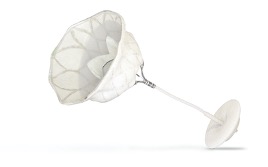
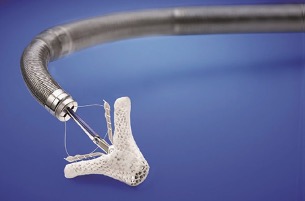
Of the four heart valves, the larger and more complex mitral valve is the most challenging to repair and replace. As a renowned heart center that excels in valve surgery, UVA has taken significant steps over the past decade to find alternative solutions to make treatment safer, easier and more effective.
UVA was involved in the first groundbreaking study that opened the door to transcatheter approaches to repair regurgitant mitral valves. “The results of the COAPT study, which we participated in and helped lead in the U.S., demonstrated marked benefit to repairing a leaking mitral valve by transcatheter approach with a device called a MitraClip™ in patients already on optimal guideline-directed medical therapy for heart failure,” says Scott Lim, MD. “This study really shifted the landscape of how we think about transcatheter options for mitral valve disease.”
Since the COAPT trial and the introduction of the MitraClip, there has been even further evolution in technological design of transcatheter mitral valve repair devices, says Lim.
UVA is making these latest discoveries available to patients through its participation in several clinical trials for next-generation transcatheter mitral valve therapies. These include:
CLASP
Edwards PASCAL TrAnScatheter Valve RePair System pivotal clinical trial (CLASP IID/IIF)
This is a prospective, multicenter, randomized, controlled pivotal trial to evaluate the safety and effectiveness of transcatheter mitral valve repair with the Edwards PASCAL Transcatheter Valve Repair System compared with Abbott MitraClip in patients with degenerative mitral regurgitation. “The Pascal system is a newer evolution of a transcatheter mitral repair device,” says Lim.
EVOQUE
An early feasibility study of the Edwards EVOQUE Transcatheter Mitral Valve Replacement System
This study investigates the replacement of a regurgitant mitral valve by the Evoque EOS valve, which is delivered from a femoral venous approach.
SUMMIT
Transcatheter mitral valve replacement with the Abbott Tendyne™ TMVR System in patients with severe mitral valve disease
The SUMMIT Trial investigates transcatheter mitral valve replacement with the Tendyne valve, and in particular focuses on patients with mitral annular calcium, who otherwise would be prohibitive risk for open surgical repair or replacement.
APOLLO
Transcatheter mitral valve replacement with the Medtronic Intrepid™ TMVR System in patients with severe symptomatic mitral regurgitation
The Apollo trial is open to patients with moderate to severe – or severe symptomatic – mitral regurgitation. It’s also open to those with moderate symptomatic mitral regurgitation combined with mitral stenosis with the presence of mitral annular calcification, or MAC.
“Around 10% of mitral valve regurgitation patients have MAC, significant scar tissue and calcium around the valve,” says Lim. “Unfortunately, these patients are very challenging to treat, either to sew in a new valve or repair the valve. So up until now, if patient had mitral regurgitation and MAC, the prognosis was dismal. We didn’t have any good options. However, since the advent of transcatheter mitral valve replacement therapy, we are now studying using less-invasive transcatheter mitral valve repair platforms in these patients, and so far the outcomes are encouraging.”
MitraClip REPAIR MR
Percutaneous MitraClip device or surgical mitral valve REpair in PAtients with PrImaRy MItral Regurgitation who are candidates for surgery (REPAIR MR)
The objective of this randomized controlled trial is to compare the clinical outcome of the MitraClip™ device versus surgical repair in patients with severe primary MR who are at moderate surgical risk and whose mitral valve has been determined to be suitable for correction by MV repair surgery.
“MitraClip was initially FDA approved for patients with degenerative mitral regurgitation at very high risk or prohibitive risk for open heart surgery,” says Lim. “Since then there has been a tremendous interest in whether there is benefit in its use in patients at lower surgical risk. We know that our surgical colleagues who are experienced in mitral valve repair get excellent results, and have really set a high bar. Therefore, we are carefully studying whether it is reasonable to offer a MitraClip repair in these patients who are lower surgical risk than what is currently approved for.”
“UVA has one of the leading centers in the U.S. for transcatheter mitral valve therapies. This has been born out by close collaboration between cardiology and cardiac surgery and headed up by myself and Dr. Irving Kron over the past 20 years,” says Lim. “This is something that truly distinguishes UVA Heart and Vascular Center.”
“At one time there were only surgical therapies for the mitral valve. There are now many options that are extremely effective,” adds Kron. “We have skilled physicians that can help plan the best approach for each patient.”
See all of the studies currently enrolling at UVA Health.
To refer a patient to UVA Heart and Vascular Center, call UVA Physician Direct at 800.552.3723.